Alzheimer’s Blood Testing: A Less Invasive, More Accessible Approach
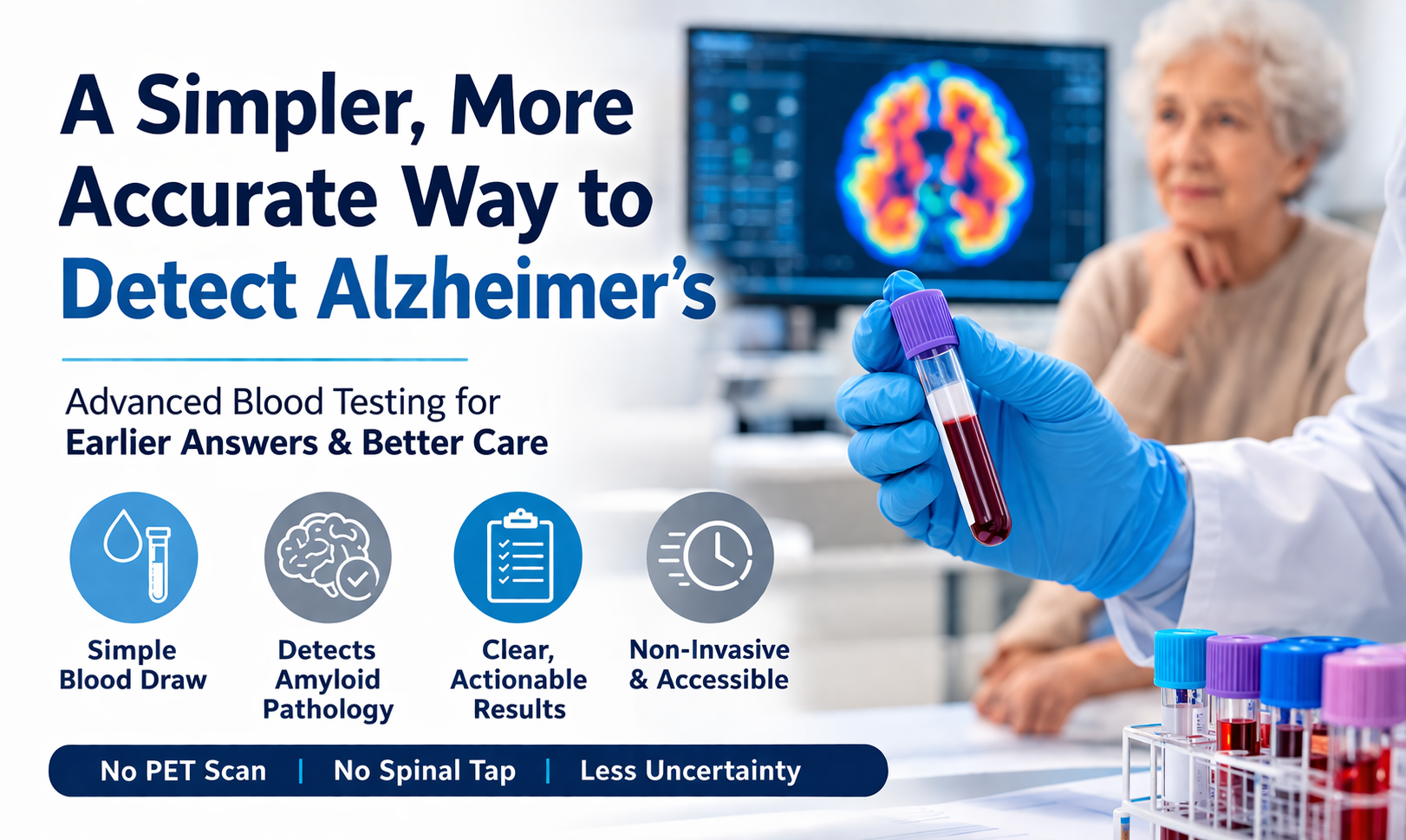
An Alzheimer’s blood test is emerging as a simpler, less invasive way to support early detection of Alzheimer’s disease. For decades, diagnosis has relied on PET imaging or cerebrospinal fluid (CSF) testing—approaches that can be costly, invasive, and difficult for many elderly patients to access.
Today, advances in biomarker science allow Alzheimer’s disease to be evaluated through a simple blood draw. With options like the LucentAD® Complete test—FDA-authorized and covered by Medicare for eligible patients aged 55 and older—providers now have a more accessible and practical way to assess patients earlier in the course of disease.
For elderly patients, particularly those in assisted living and memory care communities, these barriers can delay or even prevent proper diagnosis. Today, a new approach is changing that: the Alzheimer’s blood test.
The Rise of Blood Tests for Alzheimer’s Disease
Advancements in biomarker science have made it possible to detect Alzheimer’s disease through a simple blood draw. These blood tests for Alzheimer’s measure proteins associated with amyloid pathology—the hallmark of the disease. What once required advanced imaging or invasive procedures can now be evaluated quickly and more comfortably. This shift is transforming Alzheimer’s care by making diagnosis:
- More accessible
- Less invasive
- Better suited for elderly and high-risk populations
Why Not All Alzheimer’s Blood Tests Are the Same
While the emergence of blood-based testing is a major breakthrough, not all tests provide the same level of clarity. Many current options rely on a single biomarker, such as p-Tau217. While effective, this approach often results in a significant number of patients falling into an “indeterminate” range, where results are not clearly positive or negative.
For physicians, this creates real challenges:
- Uncertainty in diagnosis
- Delays in treatment decisions
- The need for additional testing
A More Advanced Blood Test for Alzheimer’s
At LabFlorida dba SunDx Labs, we evaluated multiple options before selecting a solution that delivers more actionable results—especially for patients in assisted living and memory care. The LucentAD® Complete blood test uses a multi-biomarker approach, analyzing five key indicators associated with Alzheimer’s disease and combining them into a single, comprehensive risk score.
The LucentAD® Complete blood test is FDA-authorized and covered by Medicare for eligible patients aged 55 and older - making it one of the few accessible, clinically validated options for early Alzheimer’s evaluation in assisted living and memory care populations.
This approach helps to:
- Reduce inconclusive results
- Improve diagnostic confidence
- Provide clear risk categories (low, intermediate, high)
For physicians, this means a more reliable tool when evaluating patients with cognitive decline.
A True Alternative to Invasive Alzheimer’s Testing
One of the most important advantages of an Alzheimer’s blood test is what it replaces. Instead of:
- Scheduling a PET scan
- Referring for a spinal tap
- Managing patient resistance or delays
Providers can now order a simple blood test.
For patients, especially older adults, this means:
- Less stress and discomfort
- No invasive procedures
- Faster access to answers
For facilities such as assisted living communities, it allows testing to be performed more easily and efficiently within the care setting.
Improving Early Detection and Patient Care
Early detection of Alzheimer’s disease is critical for:
- Care planning
- Treatment decisions
- Family communication
A blood test makes early evaluation far more practical—especially for patients who would otherwise avoid or be unable to complete traditional diagnostic procedures.
Bringing Clarity to Alzheimer’s Diagnosis
Alzheimer’s disease remains complex, and no single test replaces clinical judgment. However, better tools enable better decisions. By offering a less invasive, highly accessible alternative, blood-based biomarker testing is helping remove long-standing barriers to Alzheimer’s diagnosis.
With options like the LucentAD® Complete blood test—FDA-authorized and covered by Medicare for eligible patients aged 55 and older—providers now have a practical, reliable way to evaluate patients earlier and with greater confidence.
At LabFlorida (SunDx Labs), we are committed to bringing these advancements directly to the patients and providers we serve—especially in assisted living and memory care settings where traditional testing is often not feasible. Because when it comes to Alzheimer’s, earlier insight, clearer answers, and timely action can make a meaningful difference in patient care.